Researchers from the Hubrecht Institute in Utrecht, Erasmus MC University Medical Center Rotterdam, and Maastricht University in the Netherlands have found that the coronavirus SARS-CoV-2, which causes COVID-19, can infect cells of the intestine and multiply there. Using state-of-the-art cell culture models of the human intestine, the researchers have successfully propagated the virus in vitro, and monitored the response of the cells to the virus, providing a new cell culture model for the study of COVID-19. These findings could explain the observation that approximately one third of COVID-19 patients experience gastrointestinal symptoms such as diarrhea, and the fact that the virus often can be detected in stool samples. The results of this study were published in the scientific journal Science.
Patients with COVID-19 show a variety of symptoms associated with respiratory organs – such as coughing, sneezing, shortness of breath, and fever – and the disease is transmitted via tiny droplets that are spread mainly through coughing and sneezing. One third of the patients however also have gastrointestinal symptoms, such as nausea and diarrhea. In addition, the virus can be detected in human stool long after the respiratory symptoms have been resolved. This suggests that the virus can also spread via so-called “fecal-oral transmission”.
Intestinal organoids
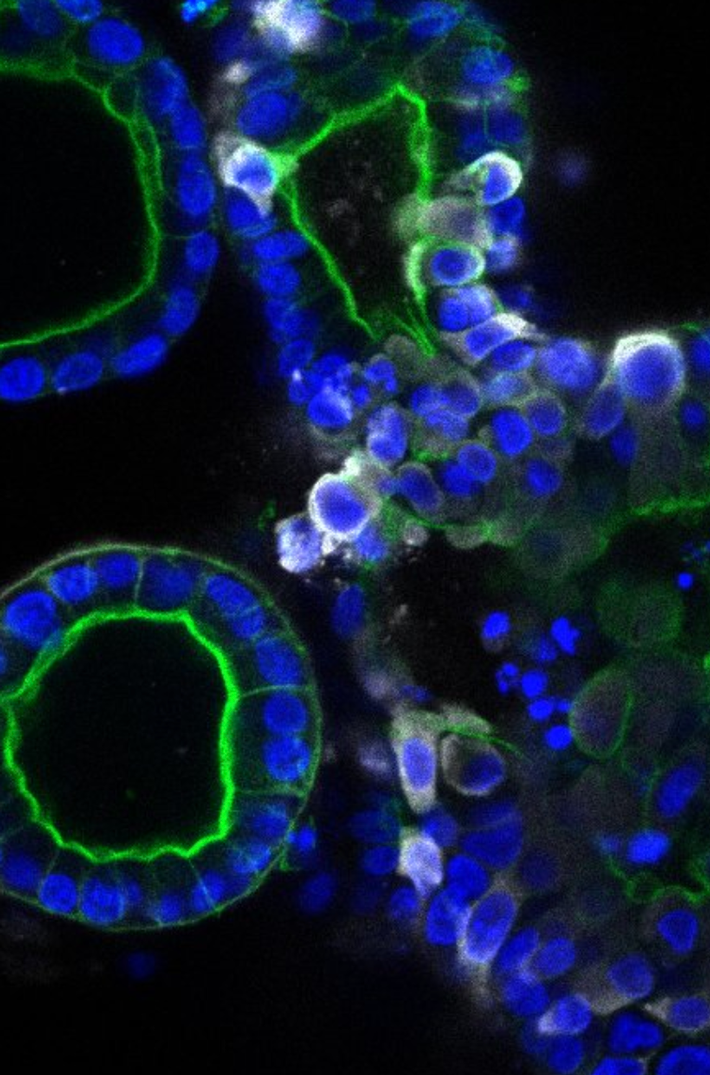
Researchers from the Hubrecht Institute, Erasmus MC and Maastricht University set out to determine whether the SARS-CoV-2 virus can directly infect the cells of the intestine, and if so, whether it can replicate there as well. They used human intestinal organoids: tiny versions of the human intestine that can be grown in the lab. Hans Clevers (Hubrecht Institute): “These organoids contain the cells of the human intestinal lining, making them a compelling model to investigate infection by SARS-CoV-2.”
Infection of intestinal cells
When the researchers added the virus to the organoids, they were rapidly infected. The virus enters a subset of the cells in the intestinal organoids, and the number of cells that are infected increases over time. Using electron microscopy, an advanced way to visualize different components of the cell in great detail, the researchers found virus particles inside and outside the cells of the organoids. Peter Peters (Maastricht University): “Due to the lockdown, we all studied virtual slides of the infected organoids remotely from home.”
The researchers also cultured the organoids in different conditions that result in cells with higher and lower levels of the ACE2 receptor, through which SARS-CoV-2 can enter the cells. To their surprise, they found that the virus infected cells with both high and low levels of the ACE2 receptor. Ultimately, these studies may lead to new ways to block the entry of the virus into our cells.
Implications
The current study is in line with other recent studies that identified gastrointestinal symptoms in a large fraction of COVID-19 patients and virus in the stool of patients free of respiratory symptoms. Special attention may be needed for those patients with gastrointestinal symptoms. More extensive testing using not only nose and throat swabs, but also rectal swabs or stool samples may thus be needed.
In the meantime, the researchers are continuing their collaboration to learn more about COVID-19. They are studying the differences between infections in the lung and the intestine by comparing lung and intestinal organoids infected with SARS-CoV-2.
Publication
Lamers et al., SARS-CoV-2 productively infects human gut enterocytes. Science 2020, 369(6499):50-54




