Replicate the success of our customer Merus:
CASE STUDY
Despite advances in systemic therapy, treatment efficacy in mCRC remains suboptimal. Standard regimens (e.g., fluoropyrimidine + oxaliplatin and/or irinotecan, often combined with targeted agents) still leave a substantial fraction of patients without meaningful benefit—while exposing them to toxicity and consuming valuable time. Additionally, attrition rates for new agents in mCRC are higher than in other indications.
Key challenges for drug discovery and translational teams include:
Patient-derived organoids capture key biological and genomic features of the original tumor and can be used for functional drug sensitivity testing. This allows you to evaluate compound activity in a model that is closer to patient biology than conventional cell lines—supporting:
PROOF OF CONCEPT
OPTIC trial: A large prospective study provides strong evidence that PDO drug response can reflect clinical outcomes in mCRC. Patients underwent metastatic biopsies for PDO establishment prior to systemic therapy. PDOs were screened with a multi-drug panel and compared to patient response.
Takeaway: PDO-based screening can help identify (in)effective therapies and strengthen confidence in translational decisions.

“Patient-Derived Organoids Predict Treatment Response in Metastatic Colorectal Cancer” (Clin Cancer Res, 2025)
We offer a practical screening setup designed for discovery and translational teams working in mCRC.
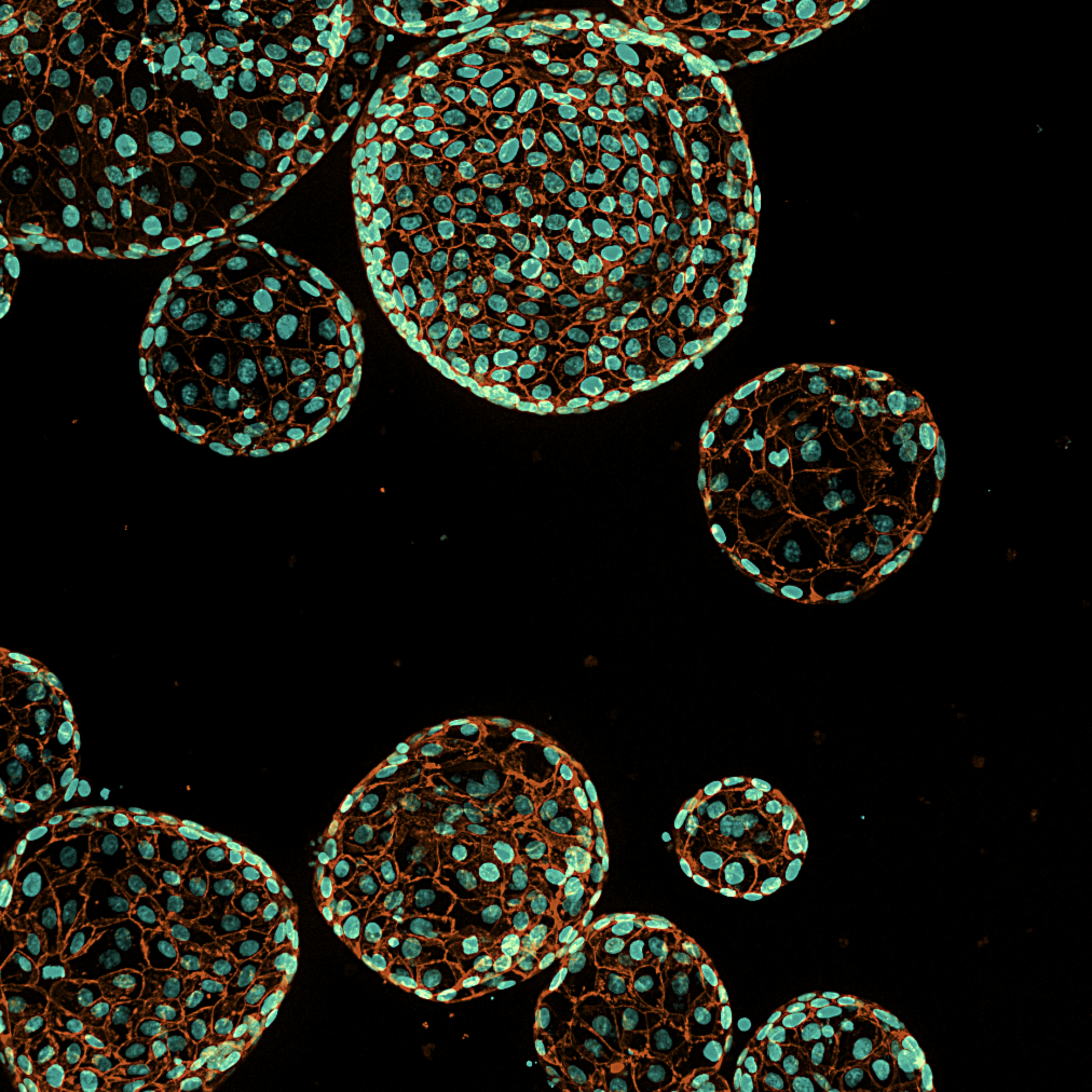
Model systems
Assay readout
What you receive (typical deliverables)
Choose the package that matches your stage and question:

3 models, up to 500 compounds (triplicates, 1 concentration)
Best for early discovery triage and identifying hits across a small, focused model set.
![]()
14 models, up to 5 compounds or 3 combinations
Best for testing lead candidates across broader biological diversity and identifying responder/non-responder patterns.
22 mCRC + 6 normal models, up to 2 compounds or 1 combination
Best for translational confidence: activity across a larger mCRC set plus normal-PDOs to identify off-target and off-tumor liabilities.

Replicate the success of our customer Merus:
CASE STUDY
Deep expertise in adult stem cell–derived organoid technology
Robust QC and reproducibility focus
Screening experience across oncology programs and modalities
Access to diverse patient-derived models and translational know-how
Clinically-relevant models to test efficacy and study resistance mechanisms
Accurately predicting treatment response in metastatic colorectal cancer (mCRC) is critical to avoid unnecessary toxicity and improve patient outcomes.
Proof-of-concept study highlighting how HUB Organoids can be used to accelerate your preclinical drug development timeline to just about five years.